Sponsors
×
[PR]上記の広告は3ヶ月以上新規記事投稿のないブログに表示されています。新しい記事を書く事で広告が消えます。
Enthalpy
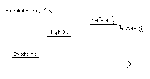
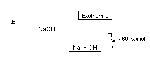
How do we mesure heat?
Joule - it takes 4.19J to head 1.0gram of water 1℃
For water the specific heat capacity is C = 4.19J/g℃
Q = mcΔT
1) how much heat is needed to heat 250 grams of water from 15℃ to 85℃ ?
memo; Q = Joule, m = mass of water (water could be other)
ΔHfor → formation
ΔHc → compustion
ΔHfus → fusion
ΔHsolid → solidification
ΔHvap → vaporization
ΔHsol → solution
ΔHneu → neutralization
ΔHdis → dissociation
ΔHrxn → reaction
ΔHion → ionization
ΔHdec → decomposition
ΔHnet → net reaction
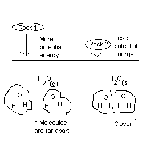
Energy Substance contains Energy
Energy Rich - CH4, H2, C3H8, C25H52, CH3OH
Energy Poor - H2O, CO2, NaCl, CaCO3
(Energy, chemical energy, enthalpy, potential energy, stored energy)
Heat, light - ?
Temperature → ℃ → 37℃ Body Tend
Heat → Joule, Calorie
Temperture - Average linetic Energy.
Heat - Sum of all energies of all molecules.
Kinetic Energy - Motion
Potential Energy - Position
Coustants:
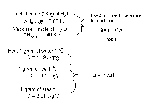
How do we mesure heat?
Joule - it takes 4.19J to head 1.0gram of water 1℃
For water the specific heat capacity is C = 4.19J/g℃
Q = mcΔT
1) how much heat is needed to heat 250 grams of water from 15℃ to 85℃ ?
| Q = ? m = 250g c = 4.18J/g℃ ΔT = 85℃-15℃ = 70℃ |
Q = (250g)(4,18J/g℃)(70℃) = 73150J Q = 73.15 KJ |
ΔHfor → formation
ΔHc → compustion
ΔHfus → fusion
ΔHsolid → solidification
ΔHvap → vaporization
ΔHsol → solution
ΔHneu → neutralization
ΔHdis → dissociation
ΔHrxn → reaction
ΔHion → ionization
ΔHdec → decomposition
ΔHnet → net reaction
Energy Substance contains Energy
Energy Rich - CH4, H2, C3H8, C25H52, CH3OH
Energy Poor - H2O, CO2, NaCl, CaCO3
(Energy, chemical energy, enthalpy, potential energy, stored energy)
Heat, light - ?
Temperature → ℃ → 37℃ Body Tend
Heat → Joule, Calorie
Temperture - Average linetic Energy.
Heat - Sum of all energies of all molecules.
Kinetic Energy - Motion
Potential Energy - Position
Coustants:
PR
