Sponsors
×
[PR]上記の広告は3ヶ月以上新規記事投稿のないブログに表示されています。新しい記事を書く事で広告が消えます。
Chemical Kinetics = the branch pf chemistry that deals with the speed of reaction and the stepwise process they follow (MECHANISM)
Reaction Mechanism
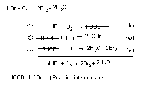
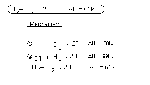
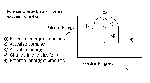
Reaction Intermediate - Those short-liyed products in a reaction mechanism that one consumed in subseqvent steps
Activation Energy - is the energy required to start a reaction by forming and activating complex
Activation complex - this is the specis or the tring formed when the reactants in a chemical reaction have collided with sufficient enrgy. to meet the activation energy requiment
Reaction mecanism - thoughs short lift products in a chemical reaction that are consumed in subsiqvent steps
Rate determenting step - that is the slowest step in reaction mechanism that determines the overl\al rate of reaction
Catalyst - a substanve that speed up a reaction but is not consumed produsing a different reaction mechanism
Rates of Chemical Reactions
Rxn Rate = Change in reaction/Products over time
Rate(rxn) = 【Amount(final) - Amount(initial)】/【Time(final) - Time(initial)】= ΔA/ΔT (Mol/s)
As a concentration:
Rate(rxn) = 【(A)final - (A)initial】/【(T)final - (T)initial】= Δ[A]/ΔT (Mol/L-s)
Kinatic Molecular Theory;
- All matter is made up of microscopic sized particles
- These particles are in constant motion (kinetic energy)
- Spaces are present between the particles of matter
- Adding heat inceases the speed of the moving particles which increases the kinetic Energy
Collision Theory:
This is simply an extension of the linetic theory
It explains that in order for a reaction to occur. Reactions particles must collide. Successfully with one another.
Factors that effect reaction Rate:
1. Temperture
2. Concentration
3. Surface area
4. Catalysts
5. Nature of Reactants
Temp: Increase in temp results in an increase in Cxn rate as mell as increase in intensity of collisions
Conc: More collisions are probable if higher concentration of particles
Surface Area: Increasing the surface area will speed up the reaction
Nature of Reactants: Simple ions + ionic compounds react faster than moleculer compounds
- Reactions that break weaker bonds react faster than those with stronger bonds
- Reactions that must break fewer bonds are faster than those with a greater number of bonds
In order for a reaction to occur, two criteria must be met.
1. Correct orientation of Particles
2. Cofficient collision energy
Activation energy: (Ea) The minimum collision energy that is requoired for a reaction to occur
Transition State Thepry: - Examines the transition from reactants to products
We can represent the increase in potential energy during a chemical reaction using a potential energy diagram (charts the potential energy of a rxn against the process of the rxn)
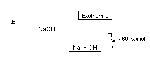
Reaction Mechanism
Reaction Intermediate - Those short-liyed products in a reaction mechanism that one consumed in subseqvent steps
Activation Energy - is the energy required to start a reaction by forming and activating complex
Activation complex - this is the specis or the tring formed when the reactants in a chemical reaction have collided with sufficient enrgy. to meet the activation energy requiment
Reaction mecanism - thoughs short lift products in a chemical reaction that are consumed in subsiqvent steps
Rate determenting step - that is the slowest step in reaction mechanism that determines the overl\al rate of reaction
Catalyst - a substanve that speed up a reaction but is not consumed produsing a different reaction mechanism
Rates of Chemical Reactions
Rxn Rate = Change in reaction/Products over time
Rate(rxn) = 【Amount(final) - Amount(initial)】/【Time(final) - Time(initial)】= ΔA/ΔT (Mol/s)
As a concentration:
Rate(rxn) = 【(A)final - (A)initial】/【(T)final - (T)initial】= Δ[A]/ΔT (Mol/L-s)
Kinatic Molecular Theory;
- All matter is made up of microscopic sized particles
- These particles are in constant motion (kinetic energy)
- Spaces are present between the particles of matter
- Adding heat inceases the speed of the moving particles which increases the kinetic Energy
Collision Theory:
This is simply an extension of the linetic theory
It explains that in order for a reaction to occur. Reactions particles must collide. Successfully with one another.
Factors that effect reaction Rate:
1. Temperture
2. Concentration
3. Surface area
4. Catalysts
5. Nature of Reactants
Temp: Increase in temp results in an increase in Cxn rate as mell as increase in intensity of collisions
Conc: More collisions are probable if higher concentration of particles
Surface Area: Increasing the surface area will speed up the reaction
Nature of Reactants: Simple ions + ionic compounds react faster than moleculer compounds
- Reactions that break weaker bonds react faster than those with stronger bonds
- Reactions that must break fewer bonds are faster than those with a greater number of bonds
In order for a reaction to occur, two criteria must be met.
1. Correct orientation of Particles
2. Cofficient collision energy
Activation energy: (Ea) The minimum collision energy that is requoired for a reaction to occur
Transition State Thepry: - Examines the transition from reactants to products
We can represent the increase in potential energy during a chemical reaction using a potential energy diagram (charts the potential energy of a rxn against the process of the rxn)
PR
