Sponsors
×
[PR]上記の広告は3ヶ月以上新規記事投稿のないブログに表示されています。新しい記事を書く事で広告が消えます。
EQUILIBRIUM
Three conditions for equilibrium
1) Balanced opposing rate
2) No loss of mass from the system (closed system)
3) No apparent change taking place
Whater at equilibrium at room temperture in a closed container.
Silver Chloride in water
Hydrogen and iodine in equilibrium with hydrogen iodine
LE CHATELER'S PRINCIPLE
When a change is imposed on a chemical system at equilibrium the system responds is a manner which tends to conteract the imposed change and establishes a new equilibrium.
If a system at equilibrium is subjected to a stress, the equilibrium will be displaced in the direction that relieves the stress.
Four conditions concerning the following reaction in a closed container
2H2(g) + O2(g) ⇔ 2H2(g)O(g) + 243 KJ
1) Concentration
What happens if the concentration of hydrogen is increased?
- More oxygen is used up. More water is formed.
The stress is the addition on hydrogen, therefore according to LCP the system wants to use up the hydrogen and shift the equilibrium to the right. When the new equilibrium is established there is more water, less of oxygen and more hydrogen.
2) Temperature
What happens when we increase temperature?
- If we increase temperature, the system responds to use up the reat, since the reverse reaction is endothermic, this reaction will be favoured therefore the result will be an increase in the concentration of water will go down.
3) Pressure
What happens when we increase pressure?
- If we increase pressure, the system responds to decrease pressure, it can accomplish this by forming fewer molecules (Avagadvo's hypothesis). In doing so, more water is formed and the concentration of hydrogen + Oxygen will go down.
2A + 3B ⇔ 4C + 1D (no change)
4) Catalyst
- The addition of a catalyst produces no change in the equilibrium concentrations because it speed up both the foward and reverse reactions by the same amount
4HCL(g) + O2(g) ⇔ 2H2O(g) + 2Cl2(g) + ENERGY
Which side is favoured by the following?
a) Add O2 - Right
b) Increase pressure - Right
c) Decrease temoerature - Right
d) Remove H2O - Right
e) Increase temoerature - Left
f) Add a catalyst - no change
g) Add HCl - Right
h) Decrease pressure - Left
i) Reducing volve of the container - Right
j) Remove Cl2 - Right
Haber Process
(to produce the maximum yield of NH3)
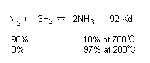
= Lower temperature is needed, must use catalyst, Favours right side,
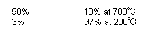
= Higher pressure is needed Favours side with fewer molecules.
Three conditions for equilibrium
1) Balanced opposing rate
2) No loss of mass from the system (closed system)
3) No apparent change taking place
Whater at equilibrium at room temperture in a closed container.
| H2O(l) + EN ⇔ H2O(g) 1) Rate of evaporation and condensation are equal 2) No water is escaping to atmosphere 3) It appears if nothing is happening |
Silver Chloride in water
| AgCl(s) ⇔ Ag+(aq) + Cl-(aq) 1) Rate of dissolving and crystalization are equal 2) No silver chloride leaves the system 3) It appears as if nothing is happening |
Hydrogen and iodine in equilibrium with hydrogen iodine
| H2 + I 2 ⇔ 2HI 2HI ⇔H2 + I 2 1) Rate of composition and decomposition the same 2) No hydrogen iodine or hydrogen iodide leaves the system (flask) 3) It appears as if nothing is happening |
LE CHATELER'S PRINCIPLE
When a change is imposed on a chemical system at equilibrium the system responds is a manner which tends to conteract the imposed change and establishes a new equilibrium.
If a system at equilibrium is subjected to a stress, the equilibrium will be displaced in the direction that relieves the stress.
Four conditions concerning the following reaction in a closed container
2H2(g) + O2(g) ⇔ 2H2(g)O(g) + 243 KJ
1) Concentration
What happens if the concentration of hydrogen is increased?
- More oxygen is used up. More water is formed.
The stress is the addition on hydrogen, therefore according to LCP the system wants to use up the hydrogen and shift the equilibrium to the right. When the new equilibrium is established there is more water, less of oxygen and more hydrogen.
2) Temperature
What happens when we increase temperature?
- If we increase temperature, the system responds to use up the reat, since the reverse reaction is endothermic, this reaction will be favoured therefore the result will be an increase in the concentration of water will go down.
3) Pressure
What happens when we increase pressure?
- If we increase pressure, the system responds to decrease pressure, it can accomplish this by forming fewer molecules (Avagadvo's hypothesis). In doing so, more water is formed and the concentration of hydrogen + Oxygen will go down.
2A + 3B ⇔ 4C + 1D (no change)
4) Catalyst
- The addition of a catalyst produces no change in the equilibrium concentrations because it speed up both the foward and reverse reactions by the same amount
4HCL(g) + O2(g) ⇔ 2H2O(g) + 2Cl2(g) + ENERGY
Which side is favoured by the following?
a) Add O2 - Right
b) Increase pressure - Right
c) Decrease temoerature - Right
d) Remove H2O - Right
e) Increase temoerature - Left
f) Add a catalyst - no change
g) Add HCl - Right
h) Decrease pressure - Left
i) Reducing volve of the container - Right
j) Remove Cl2 - Right
Haber Process
(to produce the maximum yield of NH3)
= Lower temperature is needed, must use catalyst, Favours right side,
= Higher pressure is needed Favours side with fewer molecules.
PR
