Sponsors
×
[PR]上記の広告は3ヶ月以上新規記事投稿のないブログに表示されています。新しい記事を書く事で広告が消えます。
Oxidation -reduction (Redox)
" LEO the lion says GER "
Loss of Elections = Oxidation
Gain of Elections = Reduction
- Fe0 + S0 → Fe2+S2-
- Fe0 = 26pt, 26e-
- S0 = 16pt, 16e-
pt = protons
e- = electrons
* Fe is the reducing agent.
* S is the oxidizing agent.
Na+Cl- + Ag+NO3- → Na+Na3- + Ag+Cl-
There is nothing being oxidited, nothing being reduiced. NOT oxidation-reaction.
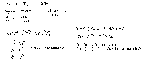
Oxidation Numbers
- The number of electrons that an atom lost or gained (or assumed) when it undergoes reaction.
* Hydrogen is a1+ except when it is paired up with something with a higher electronegativity.
# p+ = # e- → No change.
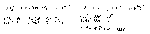
Balance the number of Electrons on each side of equition. -This is called Net ionic equition.
You may able to know the charge from given chart or periodec table.
" LEO the lion says GER "
Loss of Elections = Oxidation
Gain of Elections = Reduction
- Fe0 + S0 → Fe2+S2-
- Fe0 = 26pt, 26e-
- S0 = 16pt, 16e-
pt = protons
e- = electrons
* Fe is the reducing agent.
* S is the oxidizing agent.
Na+Cl- + Ag+NO3- → Na+Na3- + Ag+Cl-
There is nothing being oxidited, nothing being reduiced. NOT oxidation-reaction.
Oxidation Numbers
- The number of electrons that an atom lost or gained (or assumed) when it undergoes reaction.
|
H++Cl7+O42-, 8- H+Cl5+O42-, 6- H+Cl3+O24-, 2- H+Cl+O2- H+Cl- Cl ±1, 3, 5, 7 |
* Hydrogen is a1+ except when it is paired up with something with a higher electronegativity.
|
Na1+H Ba2+O2- H22+O22- O2+F22- |
|
NH3-H33+, + N22-H4+ N1-H22+, +O2-H+ |
N20 N21+O2- N2+O2- |
N23+O36-, 2- N4+O22- N25+O510-, 2- |
Balance the number of Electrons on each side of equition. -This is called Net ionic equition.
You may able to know the charge from given chart or periodec table.
PR
